Natural Variations of Isotopic Abundances
Standard atomic weights commonly are thought of as constants of nature, despite the fact that atomic weights of many common chemical elements show variations as a result of physical, chemical and biological processes. For example, the atomic weight of boron can be anywhere between 10.806 and 10.821, depending on where the element is found.
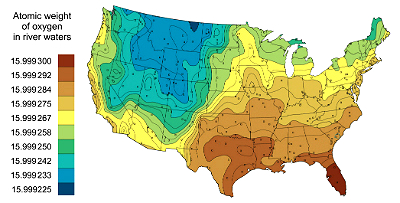
Variation in atomic weight of oxygen in river waters across the continental United States (Holden et al 2018, Kendall and Coplen 2001)
For twelve elements, CIAAW has compiled published data showing the variation of isotopic composition and atomic weight in various materials. Since 2009, these plots are published by CIAAW in its biennial reports.
CIAAW SNIF diagrams
Citation
T.B. Coplen and Y. Shrestha. Isotope-abundance variations and atomic weights of selected elements: 2016 Pure Appl. Chem. 88, 1203-1224 (2017)
For more information please consult other publications by CIAAW.


